The ADA Forsyth Institute’s Core Facilities and Services provides specialized equipment, training and expertise on a fee-for-service basis to researchers from external institutions. AFI’s state-of-the-art facilities, advanced technologies, and experienced core managers can accommodate your project needs.
The ADA Forsyth Institute Advanced Microscopy Core maintains four light microscopes listed below for use by interested researchers.
Services
- Instrument training
- Independent access to instruments for trained users
- Assisted use of instruments with help of experienced microscopist
- Consultations for experiment design, sample prep, imaging strategy, image analysis, and writing methods for publications
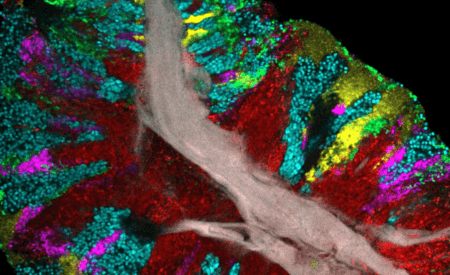
- Accommodations for many sample types and imaging techniques including:
- Imaging cleared tissue with CLARITY system
- Zeiss Airyscan for fast, high-resolution imaging
- Time lapse and live cell imaging
- Bacteria labelling with fluorescence in situ hybridization (FISH)
- Multiplexed confocal imaging of 10 or more fluorophores simultaneously
- Near-infrared detection
- Imaging large tissue sections with epifluorescence, confocal, or color imaging
Equipment
Leica Thunder Imager Model Organism (TIMO)
 Fluorescence stereomicroscope ideal for imaging large specimens
Fluorescence stereomicroscope ideal for imaging large specimens
- Allows fast and easy 3D exploration of large samples including whole organisms
- Thunder provides image quality improvement by removing out of focus blur
- 3 Fluorescence filter sets (blue, green, red)
- Transmitted light and incident light options for observing the sample
- Automated focus column to collect Z-stacks easily
- Equipped with highly sensitive sCMOS camera
Zeiss Axio Observer inverted microscope

Ideal for labeled or unlabeled samples, live cell imaging, and tile scanning of large tissues.
- Inverted, fully automated microscope
- Transmitted light techniques to visualize any sample (brightfield, DIC, phase contrast, polarized light)
- Perform epifluorescence using Colibri 2 LEDs and 4 filter sets (blue, green, red, far-red)
- Image with sCMOS camera for speed and high sensitivity
- Color camera for light absorbing dyes (eg. H&E)
- Perform live cell experiments with stage-top incubation system for temperature and CO2 control
- Image over large areas with fast tile scanning
Zeiss LSM 980 confocal inverted microscope

Acquired with an NIH S10 award and installed in 2023, this is our facility’s workhorse.
- Excitation flexibility with 8 solid-state laser lines
- Detection flexibility with up to 36 simultaneous detector channels from 400-900nm including Near Infrared
- Image more than 10 fluorophores simultaneously with linear unmixing
- LSM Plus for improvement in signal and resolution
- AI Sample Finder for quick and easy sample location and navigation
- Zen Connect for correlating images across scales and modalities
- Collect Z-stacks for visualization and analysis of 3D structures
Zeiss LSM 880 confocal upright microscope
 Ideal for imaging cleared samples and for gaining higher resolution
Ideal for imaging cleared samples and for gaining higher resolution
- Excitation flexibility with 7 laser lines
- Detection flexibility between 400-800nm
- Airyscan detector for super resolution and faster imaging
- 1.7x higher resolution and 4-8x higher signal compared to traditional confocal
- FAST mode for 4x faster acquisition
- 20x/1.0 dipping objective for imaging cleared specimens (RI=1.45), eg. CLARITY
- Temperature housing
- Collect Z-stacks for visualization and analysis of 3D structures
Image Analysis
The Core provides access to an offline workstation equipped with Imaris Microscopy Image Analysis Software and other open source software for conducting image processing and analysis.
Leica Thunder Imager Model Organism (TIMO)
Zeiss Axio Observer inverted microscope
Zeiss LSM 980 confocal inverted microscope
Zeiss LSM 880 confocal upright microscope
Pricing
| Instrument/Service | Academic Rate | Commercial Rate |
| Confocal microscopes (Zeiss LSMs) | $100 / hr | $125 / hr |
| Widefield (Zeiss Axio Observer) and Stereomicroscope (Leica TIMO) | $60 / hr | $75 / hr |
| Training/Assisted Use | $80 / hr | $100 / hr |

Advanced Microscopy Core Services
Advanced Microscopy Core Manager Jennifer Gundrum has over seven years of experience in advanced imaging services. Contact her for inquiries about services.
